Naturally occurring isotopes are marked in green light green = naturally occurring radionuclides. 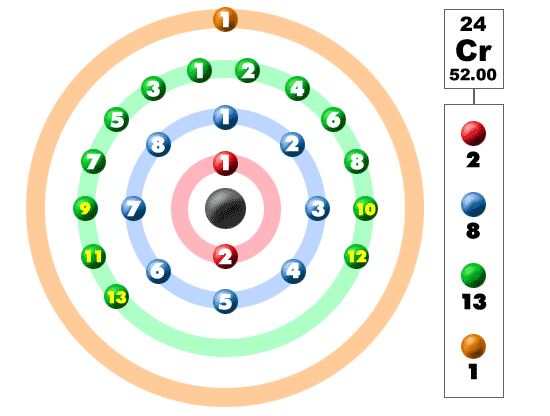
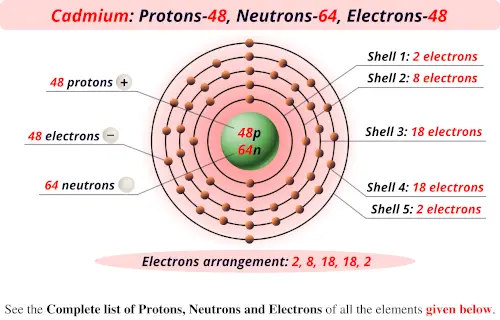
Isotones and IsobarsThe following table shows the atomic nuclei that are isotonic (same neutron number N = 39) and isobaric (same nucleon number A = 63) with Chromium-63. Half-life T ½ = 129(2) ms respectively 1.29 × 10 -1 seconds s. Take the mass number for each element above and subtract the atomic number (number of protons). The atomic number (usually the whole number.increases one after the other), is the number of protons. Chromium has an atomic number of 24, so it always has 24 protons. See Below Look up each element on the periodic table. To determine the number of protons, electrons, and neutrons in these elements, we must first understand that the atomic number of an element, which is the number of protons in the nucleus of an atom of that element, is constant. 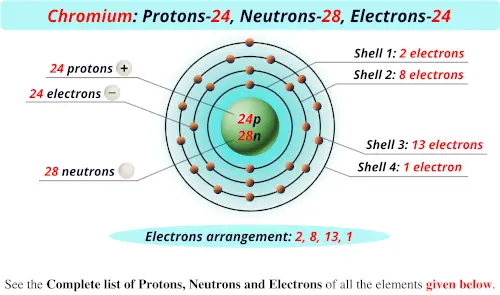
The 58 is the mass of the Chromium isotope. Chromium metal is valued for its high corrosion resistance and hardness. (Protons Electrons) Chromium-58 Chromium-63 of protons 24 24 of neutrons of electrons 24 24 3) To find the of neutrons, you would take the mass of the isotope and subtract the number of protons. It is a steely-grey, lustrous, hard, and brittle transition metal. Question: Determine the number of protons, electrons, and neutrons for the isotope chromium-58. 58 Ce Cerium 140.12 59 Pr Praseodymium 140.91 60 Nd Neodymium 144.24 61 Pm. 15 hours ago &0183 &32 It is neutron rich, containing 82 protons and 126 neutrons. Name of the isotope: Chromium-63 Cr-63 Symbol: 63Cr or 63 24Cr Mass number A: 63 (= number of nucleons) Atomic number Z: 24 (= number of protons) Neutrons N: 39 Isotopic mass: 62.96134(38) u ( atomic weight of Chromium-63) Nuclide mass: 62.9481744 u (calculated nuclear mass without electrons) Mass excess: -36.01156 MeV Mass defect: 0.564392492 u (per nucleus) Nuclear binding energy: 525.72821994 MeV (per nucleus) 8.34489238 MeV (average binding energy per nucleon) Separation energy: S N = 3.18(39) MeV (first neutron) S P = 17.82(47) MeV (first proton) Half-life: 129(2) ms Decay constant λ: 5.373233957829 s -1 Specific activity α: 5.219092220555 × 10 +22 Bq g -1 1410565465014.9 Ci g -1 Spin and parity: (nuclear angular momentum) 1/2- Year of discovery: 1992 The given elements are Chromium 58 and Chromium 63. Chromium is a chemical element it has symbol Cr and atomic number 24. 24 Cr Chromium 51.996 25 Mn Manganese 54.938 26 Fe Iron 55.845 27 Co Cobalt.
0 Comments
Leave a Reply. |
AuthorWrite something about yourself. No need to be fancy, just an overview. ArchivesCategories |
 RSS Feed
RSS Feed
